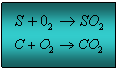Chemcial Composition
The composition of fireworks is based on a very simple theory of combustion. The composition must include 6 vital ingredients which include, fuel source, binder, oxidiser, chemical element for colour, regulator and reducing agent.
The most common fuel known in the pyrotechnic industry is black powder or gunpowder, it includes organic elements which are charcoal and thermite. The use of the oxidiser is to produce the oxygen needed to burn the fireworks. Many oxidisers include nitrates, chlorates and perchlorates. Nitrates give up a third of their oxygen which reduces the effect of the firework, whereas chlorates release all their oxygen to produce a better effect. Perchlorates contain the most oxygen but less likely to explode like the chlorates due to their stability. The next part of a firework is the reducing agent which burn the oxygen from the oxidiser to produce hot gases, common reducing agent include sulphur and charcoal. Regulators (metals) can be added to a firework to regulate the speed of the reaction of the firework. Colouring agents also known as chemical elements are used to produce different firework colours.
The most common fuel known in the pyrotechnic industry is black powder or gunpowder, it includes organic elements which are charcoal and thermite. The use of the oxidiser is to produce the oxygen needed to burn the fireworks. Many oxidisers include nitrates, chlorates and perchlorates. Nitrates give up a third of their oxygen which reduces the effect of the firework, whereas chlorates release all their oxygen to produce a better effect. Perchlorates contain the most oxygen but less likely to explode like the chlorates due to their stability. The next part of a firework is the reducing agent which burn the oxygen from the oxidiser to produce hot gases, common reducing agent include sulphur and charcoal. Regulators (metals) can be added to a firework to regulate the speed of the reaction of the firework. Colouring agents also known as chemical elements are used to produce different firework colours.
To produce the red explosion you will need to fill the stars of the firework with strontium carbonate, another common salt is lithium.
To produce the green explosion you will need to fill the stars of the firework with barium compound + chlorine produce.
To produce the blue explosion you will need to fill the stars of the firework with copper compound + chlorine produce.
To produce the yellow explosion you will need to fill the stars of the firework with sodium nitrate.
To produce the purple explosion you will need to fill the stars of the firework with both copper and strontium compounds.
To produce the gold explosion you will need to fill the stars of the firework with charcoal and it needs to be heated up to produce the incandescent light.
To produce the silver explosion you will need to fill the stars of the firework with aluminium or magnesium, both burning.
The very last ingredient which finishes the firework is the binder. Binder is a type of paste which essentially holds the firework mixture together. The most common binder is called dextrin. The effects of dextrin doesn't begin to work until the firework has been lit.
To produce the green explosion you will need to fill the stars of the firework with barium compound + chlorine produce.
To produce the blue explosion you will need to fill the stars of the firework with copper compound + chlorine produce.
To produce the yellow explosion you will need to fill the stars of the firework with sodium nitrate.
To produce the purple explosion you will need to fill the stars of the firework with both copper and strontium compounds.
To produce the gold explosion you will need to fill the stars of the firework with charcoal and it needs to be heated up to produce the incandescent light.
To produce the silver explosion you will need to fill the stars of the firework with aluminium or magnesium, both burning.
The very last ingredient which finishes the firework is the binder. Binder is a type of paste which essentially holds the firework mixture together. The most common binder is called dextrin. The effects of dextrin doesn't begin to work until the firework has been lit.
What is a Chemical Reaction?
A chemical reaction is the process that leads towards the transformation of a set of chemcial substances to another.
https://en.m.wikipedia.org/wiki/Chemical_reaction
https://en.m.wikipedia.org/wiki/Chemical_reaction
Chemical Reactions
An exploding firework is a number of chemical reactions happening spontaneously and in a rapid sequence. When you provide heat and activation energy, this makes a solid chemical compound pack within the cardboard and combusts with oxygen in the air. This releases smoke and exhaust gases including carbon monoxide, carbon dioxide and nitrogen.
Here is a chemical equation for the reaction of when the main gunpowder charge burns to ignition;
2KNO3 (potassium nitrate) + S (sulfur) + 3C (carbon in charcoal form) → K2S (potassium sulfide) + N2 (nitrogen gas) + 3CO2 (carbon dioxide)
To begin, 2KNO3 (potassium nitrate) is one of the major constituents for gunpowder, the potassium nitrate burns to create oxygen. 3C (carbon in charcoal form) is the produce of heat by slow pyrolysis, the carbon and oxygen from the potassium nitrate chemically bind to create carbon dioxide. S (Sulfur) is the main ingredient to gunpowder and is important for the burn of the gunpowder. K2S (potassium sulfide) is formed by gunpowder and the effects include the glitter formations. N2 (nitrogen gas) and the . 3CO2 (carbon dioxide) are both released when when the compound pack combusts with oxygen in the air.
Here is a chemical equation for the reaction of when the main gunpowder charge burns to ignition;
2KNO3 (potassium nitrate) + S (sulfur) + 3C (carbon in charcoal form) → K2S (potassium sulfide) + N2 (nitrogen gas) + 3CO2 (carbon dioxide)
To begin, 2KNO3 (potassium nitrate) is one of the major constituents for gunpowder, the potassium nitrate burns to create oxygen. 3C (carbon in charcoal form) is the produce of heat by slow pyrolysis, the carbon and oxygen from the potassium nitrate chemically bind to create carbon dioxide. S (Sulfur) is the main ingredient to gunpowder and is important for the burn of the gunpowder. K2S (potassium sulfide) is formed by gunpowder and the effects include the glitter formations. N2 (nitrogen gas) and the . 3CO2 (carbon dioxide) are both released when when the compound pack combusts with oxygen in the air.
|
Nitrates only give up a third of the oxygen as the equation to the left says.
On the contrary chlorates get completely reduced as they are an better oxidising agent and so causes a better reaction. These can be highly explosive.
Percholorates contain even more oxygen but are less likely to explode like the chlorates due to the increase of stabability. This follows the chemical equation reaction to the left.
This reducing agent burns the oxygen produced from the oxidisers above to produce hot gases. Common reducing agents include sulphur and charcoal. Both the reducing agents react with the oxygen to create sulphur dioxide and carbon as illustrated on the left.
|
What is a Chemical Change?
A chemcial change is any change that results in the formation of new chemical substances.
http://antoine.frostburg.edu/chem/senese/101/matter/faq/physical-chemical.shtml
http://antoine.frostburg.edu/chem/senese/101/matter/faq/physical-chemical.shtml
Chemical Change
When the time fuse on a firework is lit, the gunpowder begins to change into a flame. When the heat energy becomes too much for the mortar tube it shoots up into the sky. When the time fuse reaches the chemical compound pack it reacts and builds up pressure inside the firework. When the highest pressure is reached the firework explodes. This is an example of a chemical change.